About Us
Our Mission
To discover the mechanisms that govern the homeostasis of the diverse cell types in adipose tissue, which significantly impact the development of metabolic disease.
Our Research
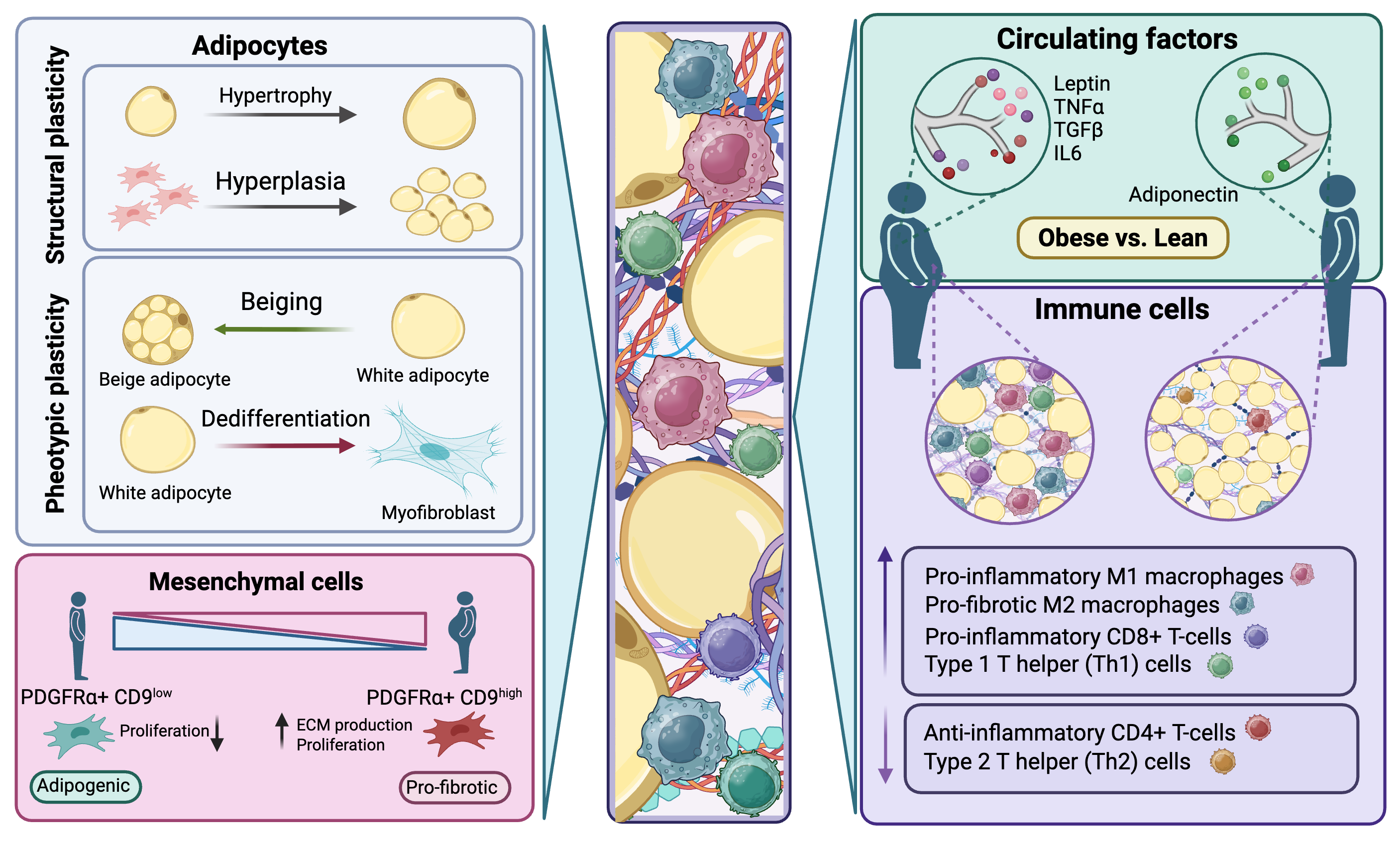
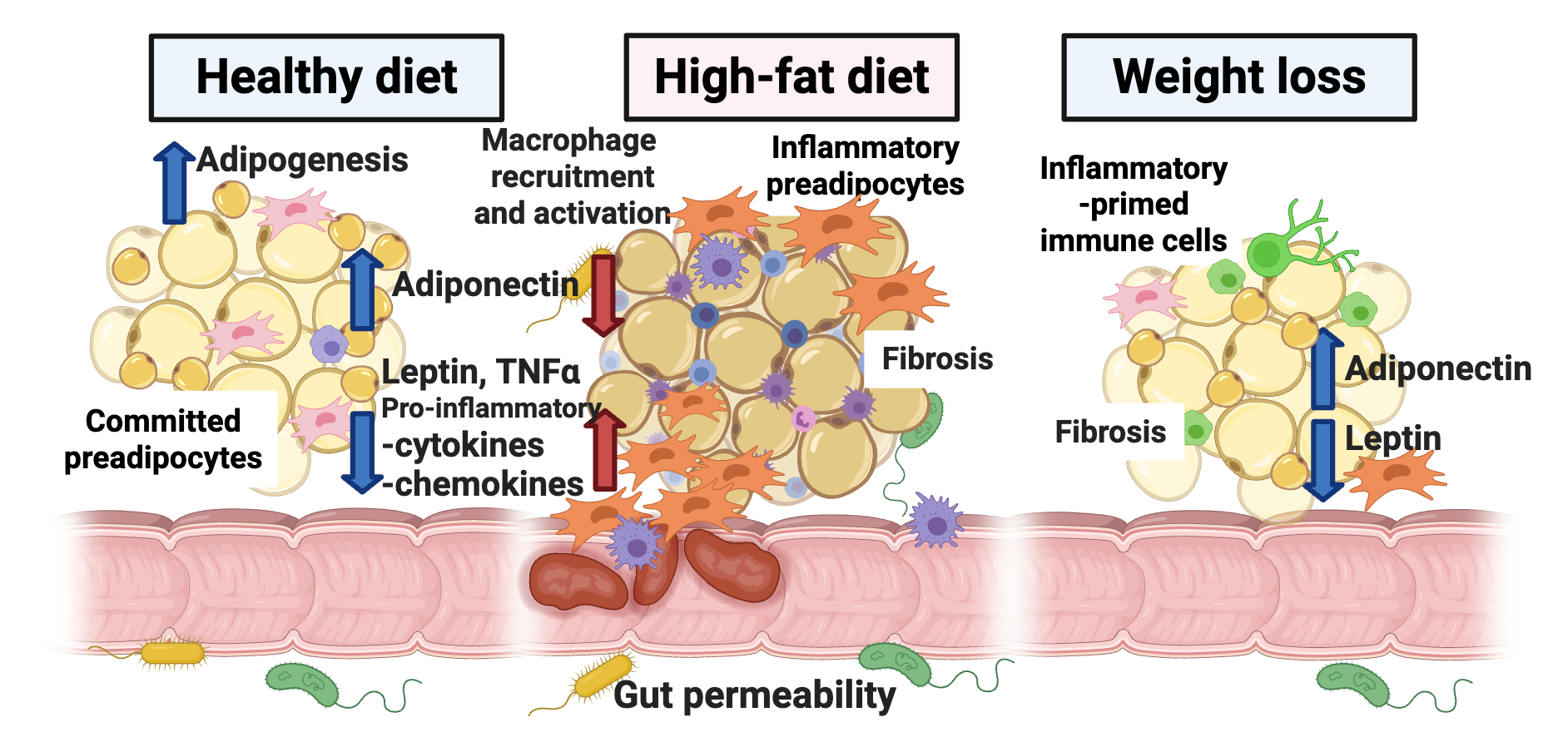
Adipose tissue is more than just “fat”—it’s an endocrine organ that responds dynamically to extracellular signals and plays a central role in energy homeostasis and immune regulation. Adipose tissue is not merely a storage reservoir. It is an active endocrine organ, secreting numerous adipokines, including hormones and cytokines, that regulate systemic glucose homeostasis, lipid metabolism, inflammation, and many other physiological events. A high-fat diet (HFD) and adipose tissue inflammation are linked to whole-body metabolic disturbances, as other organs cope with toxic levels of lipid, which would otherwise be safely stored in adipocytes. Furthermore, the anatomical location of accumulated fat, such as visceral adiposity in the intra-abdominal region, is a clear determinant of significant risk for metabolic disease.
Among fat depots, mesenteric visceral fat is strongly linked to obesity-related diseases and inflammatory bowel disease, yet it remains one of the least studied. Its hidden location in the abdomen and complex mix of cell types make it a scientific challenge, but also an opportunity to better understand why unhealthy obesity leads to widespread metabolic disease. We explore how this type of adipose tissue remodels, both in healthy and unhealthy ways, in response to overnutrition, focusing on the influence of immune cells in fat and potential therapeutic strategies to restore healthy function.
Current Projects:
Defining how mesenteric fat remodels during obesity, weight loss, and weight regain.
Our goal is to understand the cellular landscape in MAT from adipocytes to immune cells and more. We will use lineage-tracing mouse models and single-cell and nuclear RNA sequencing to overcome the heterogeneous nature of this adipose tissue.
Determine the importance of mesenteric adipose tissue inflammation to the progression of IBD.
In several well-established mouse models of Crohn’s disease (which cause ileocolitis), the gastrointestinal tract has been the primary focus. In contrast, we will focus on MAT biology and the role of mesenteric adipocytes, adipocyte progenitors, and immune cells at the intestinal interface during ileocolitis. To study how MAT influences the progression of disease, we will use mouse models that manipulate the pro- or anti-inflammatory state of MAT.